EU CLP
Regulatory Introduction
The CLP Regulation Classification, Labeling and Packaging is based on the United Nations Globally Harmonized System of Classification and Labeling (GHS), the Regulation (EC) No 1272/2008 on Classification, Labeling and Packaging of Substances and Mixtures, commonly known as the CLP Regulation, which came into effect on January 20, 2009.
The implementation of this regulation signifies that in addition to responding to REACH regulations, enterprises also need to comply with the requirements of CLP regulations in order to smoothly carry out trade with Europe. According to Article 4 of the CLP regulation, if the substance and mixture (formulation) do not meet the requirements of the CLP regulation, they will not be placed on the EU market.
The classification, labeling, and packaging (CLP) of three new hazardous substances and mixtures will take effect from April 20, 2023.
New hazard classification types
• ED HH Class 1 and Class 2 (Endocrine disruptions to human health)
• ED ENV Class 1 and Class 2 Environmental Assessment (Endocrine Disruption to the Environment)
• PBT (persistent, bioaccumulative, toxic), vPvB (very persistent, very bioaccumulative)
• PMT (persistent, mobile, toxic), vPvM (very persistent, very mobile)
The new hazard classification will be included in the IT tool IUCLID in the spring of 2024. At that time, companies will be able to include information related to new hazard classifications in their CLP notifications, REACH registrations, and Product and Process Research and Development (PPORD) notifications, as well as in their documents submitted under the European Union Biocide Regulation (BPR) and the Center for Toxicity (PCN) notifications.
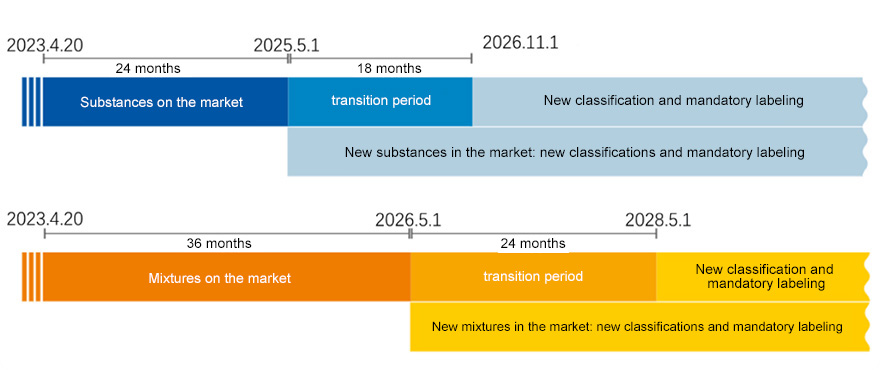
Application Date and Response Transition Period
The new regulations will come into effect on April 20, 2023. From now on, member states can propose coordinated classification and labeling (CLH) recommendations for new hazard categories, and manufacturers, importers, downstream users, and distributors can accordingly self classify their substances and mixtures. After the authorization regulations come into effect, there is a transitional period during which manufacturers, importers, downstream users, and distributors have not yet been required to classify their substances or mixtures according to new hazard categories. During this period, new hazard levels can be voluntarily applied. At the end of the transition period, all manufacturers, importers, downstream users, and distributors must adopt the new hazard level.
1、Substance registration
New substances for sale on the market: effective from May 1, 2025。
Chemical substances already on the market for sale before this date will not be required to comply with new hazard classification and labeling regulations until November 1, 2026。
2、Mixture registration
New mixtures for sale on the market: Starting from May 1, 2026, new hazard classification and labeling requirements will be implemented。
Mixtures already on the market for sale before this date are not required to comply with new hazard classification and labeling regulations until November 1, 2028。
Requirements for enterprises after the implementation of CLP regulations
― Data security table(SDS):
SDS is a comprehensive explanatory document provided by hazardous chemical production or sales enterprises to customers in accordance with regulatory requirements, which includes information on chemical composition, physical and chemical parameters, combustion and explosion performance, toxicity, environmental hazards, as well as safety use methods, storage conditions, leakage emergency response, transportation regulatory requirements, etc. It is also one of the mandatory information transmission carriers required by the EU REACH regulations.
― Classification Label (C&L) Notification:
• Notification subject: Manufacturers and importers of substances, mixed substances within the European Union
• Notification time: Within one month after the initial launch of the substance on the market, the applicant shall notify ECHA of its classification and labeling (C&L) obligations. The notification information will be entered into the public classification and labeling list, and the information will be available to the public on the ECHA website.
• Notification method: Submit notification file through REACH-IT
• Notification of substances: substances registered and marketed in accordance with REACH regulations (≥ 1 ton/year); Substances classified as hazardous under CLP and put on the market; A substance classified as hazardous under CLP and present in a mixture with a concentration limit higher than that specified in Annex I of CLP, resulting in the mixture being classified as hazardous, as well as the place where the mixture is placed on the market.
• Information to be submitted for notification: importer, manufacturer information; Material characteristics, hazard classification and labeling; Concentration limits that result in the mixture being classified as hazardous
Zhongbang Group has 12 years of technological accumulation and accumulation, with rich experience in chemical regulatory response, risk assessment technology and experience, as well as high-quality cooperative laboratory resources, to ensure service quality and efficiency, and provide comprehensive protection for cross-border business.
Labeling and Packaging
In CLP, a complete label should contain at least 5 parts: signal words, danger instructions, danger pictograms, prevention instructions, supplementary information, etc.
The Safety Data Sheet (SDS) for the unified disclosure of chemical hazard information includes the following content
Services provided
- • C&L Notification
- • Notification from the EU Toxicology Center
- • Label design
- • SDS production in compliance with CLP regulations
- • An international team provides multilingual (Chinese, English, Japanese) services
COC REACH LIMITED specializes in product registration and compliance consulting services. Leveraging its professional expertise, diverse resources, and global network, it is committed to providing regulatory consulting and environmental compliance services to pharmaceutical and chemical companies, consumer goods manufacturers, and large multinational corporations. It addresses issues related to environmental health and safety laws, product quality standards, and other challenges faced by enterprises during production, sales, and global trade processes.
Accumulated Customers
18,000+
Distributed in 35 countries and regions
15year
Industry experience
260+
Senior expert team
15+
Subsidiaries
300+
Collaboration Cases
Is it necessary to inform the list of non-hazardous substances that are also registered under REACH?
Yes, it is. Article 39(a) of the CLP states that "substances registered under Regulation (EC) ...
When is the deadline for submitting the list of categories and labels?
The deadline for submitting a notification to the list is one month after a substance is place...
Is notification limited to the production or import of one tonne or more per year?
No, in accordance with Article 39 (b) of the CLP, it is required to report that the stock incl...
What are the requirements of the EU CLP regulations for the export of products?
Before placing chemicals on the EU market, the industry must determine the potential risks to ...